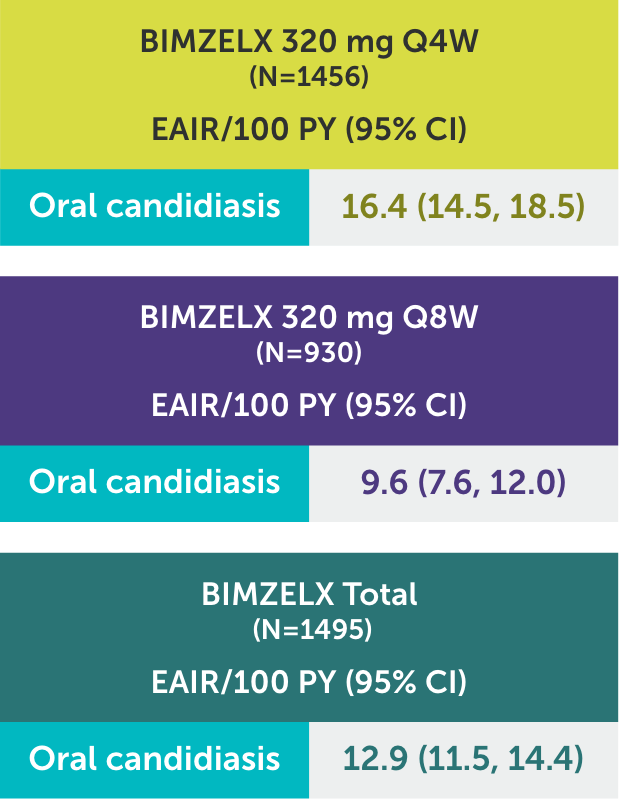
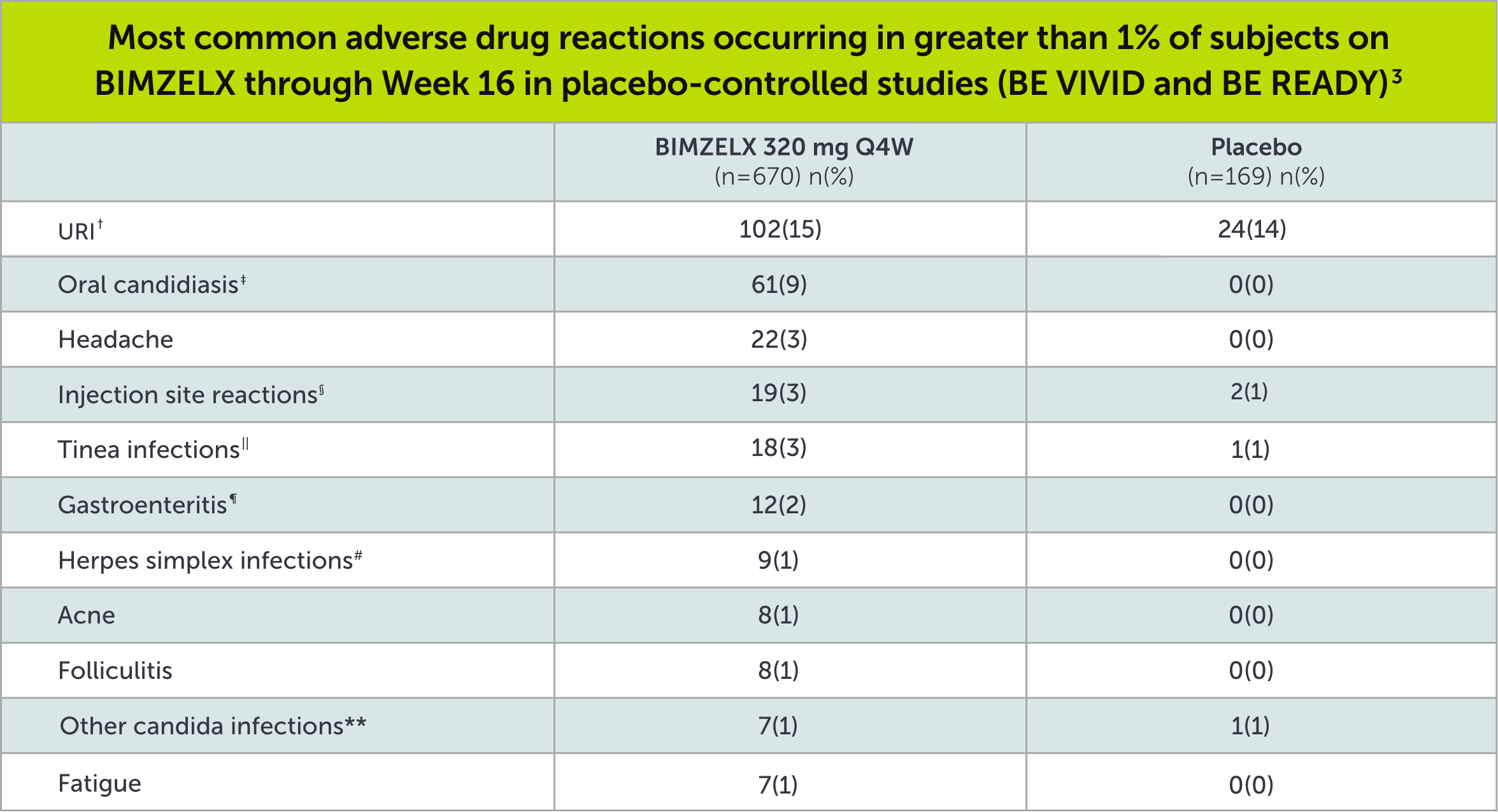
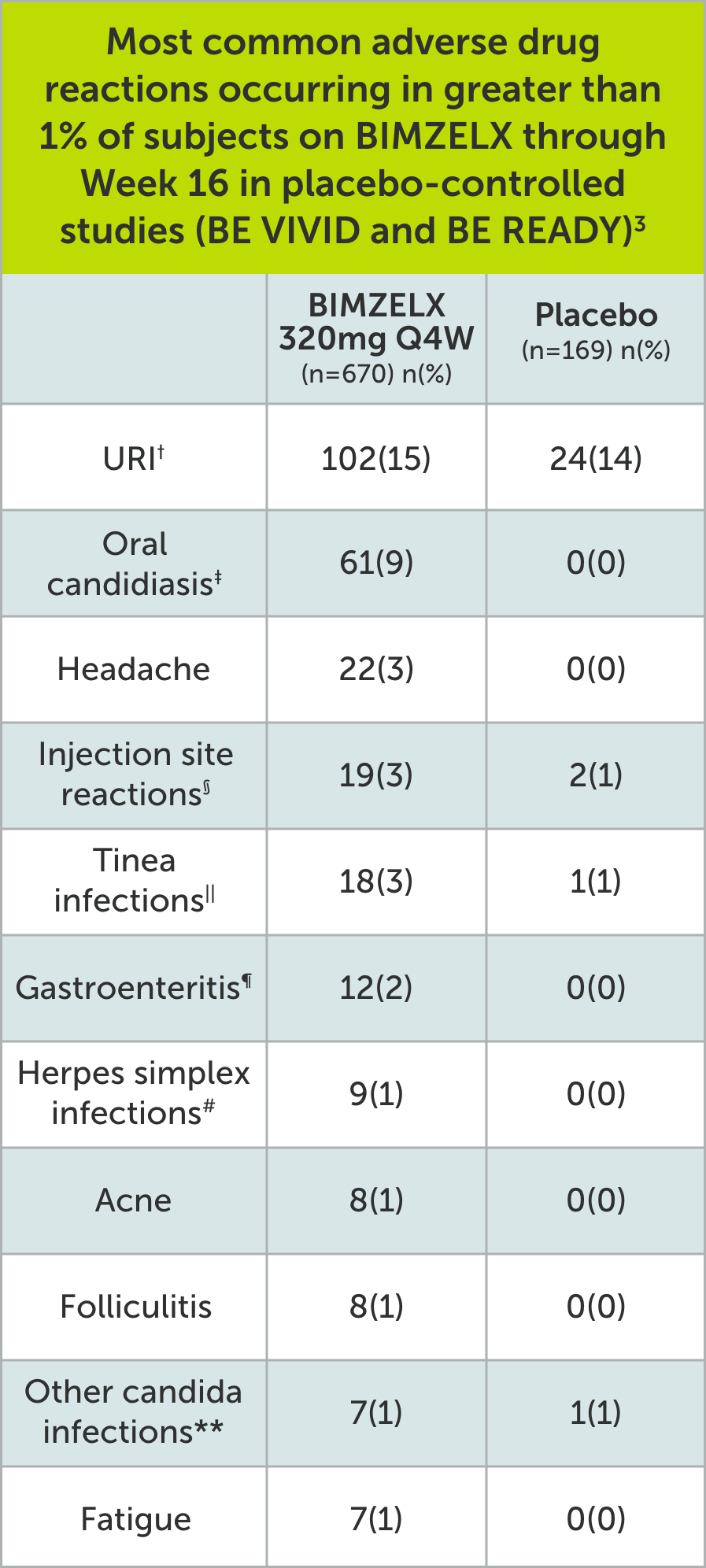
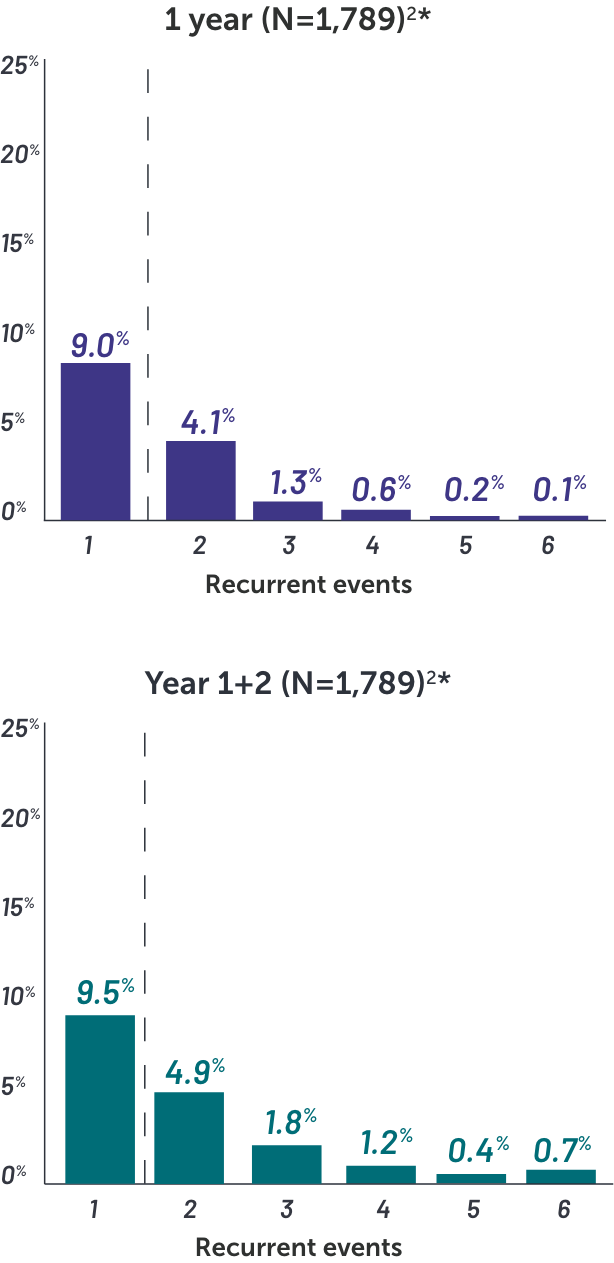SAFETY PROFILE3
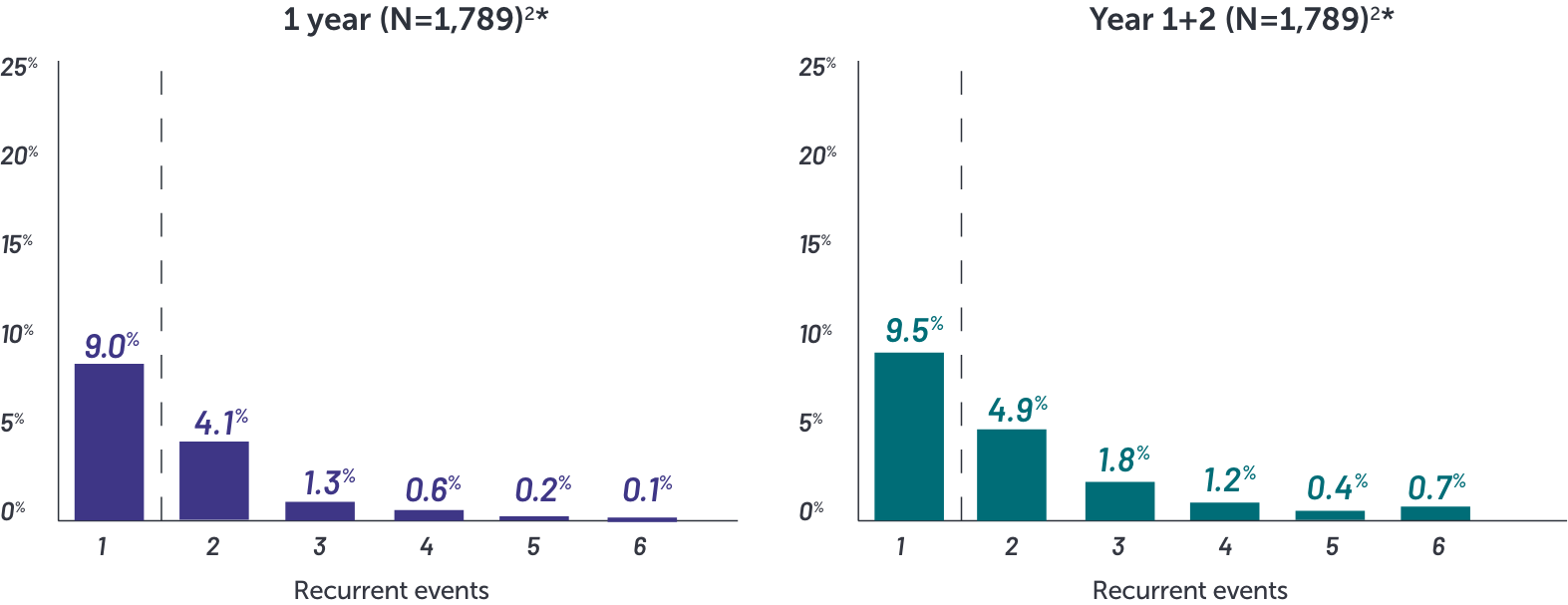
In BIMZELX clinical studies, the vast majority of oral candidiasis cases:
•Were mild to moderate1
•Resolved with standard therapy1
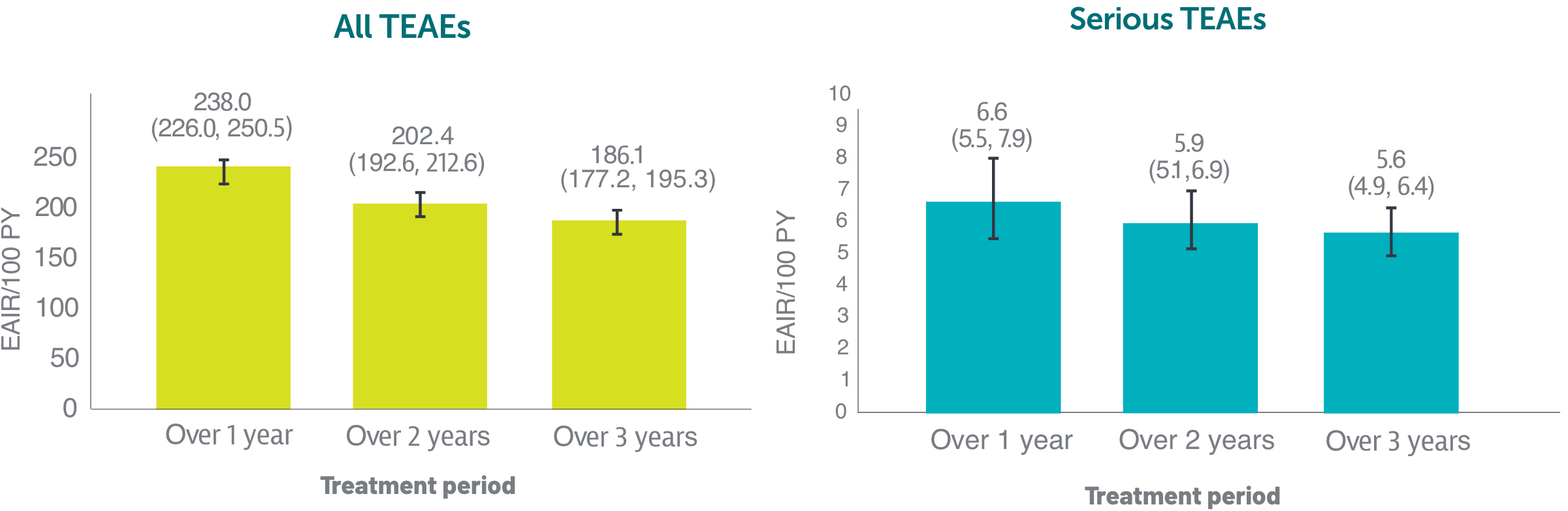
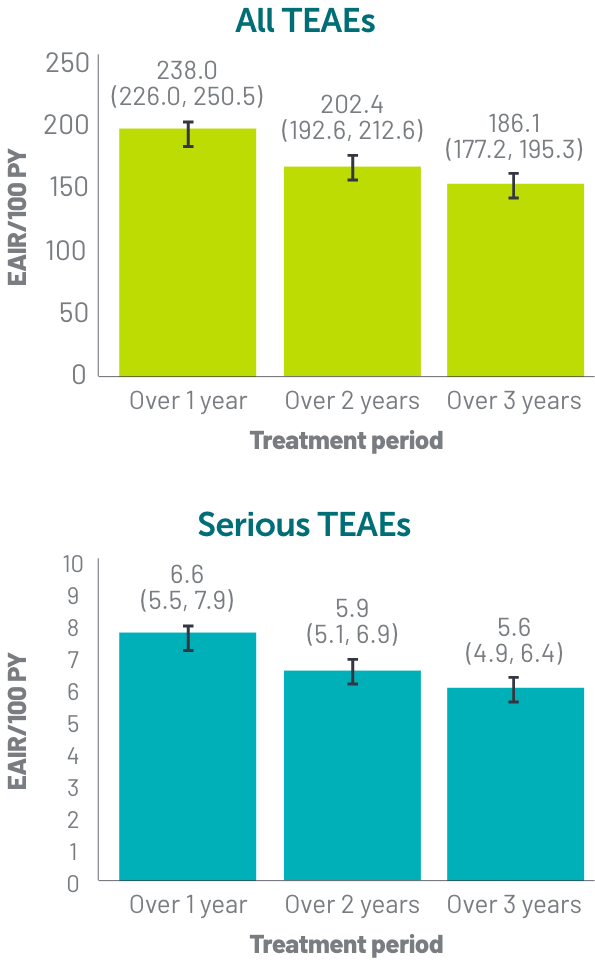
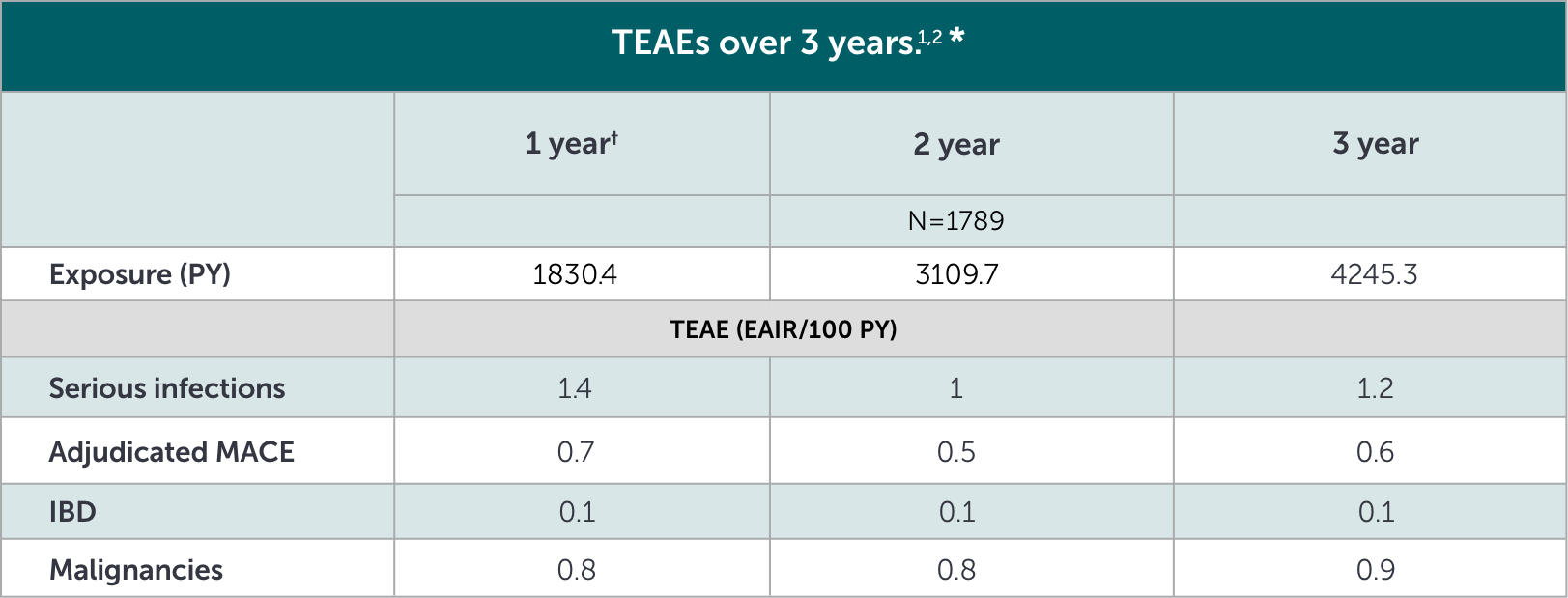
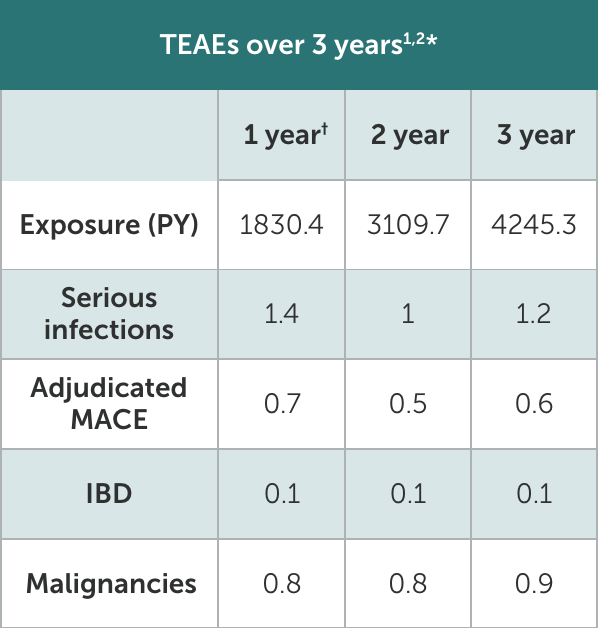
*Based on phase 2 and phase 3 studies. BE RADIANT was not included in this analysis, and adverse events in BE RADIANT were similar to those of the other clinical trials.
†Upper respiratory infections include nasopharyngitis, upper respiratory tract infection, pharyngitis, rhinitis, viral upper respiratory tract infection, tonsillitis, sinusitis, pharyngitis streptococcal, pharyngitis bacterial, peritonsillar abscess, viral rhinitis, and influenza.
‡Oral candidiasis includes oral candidiasis, oropharyngeal candidiasis, oral fungal infection, fungal pharyngitis, and oropharyngitis fungal.
§Infection site reactions include injection site reaction, injection site erythema, injection site pain, injection site edema, injection site bruising, and injection site swelling.
||Tinea infections include tinea pedis, fungal skin infection, tinea versicolor, tinea cruris, tinea infection, body tinea, and onychomycosis.
¶Gastroenteritis includes Enterovirus infection, gastroenteritis, gastroenteritis bacterial, and gastroenteritis viral.
#Herpes simplex infections include herpes simplex and oral herpes.
**Other candida infections include vulvovaginal candidiasis, vulvovaginal mycotic infection, skin candida, and genital candidiasis.
Q4W=every 4 weeks; URI=upper respiratory infection.

HELP CLEAR THE WAY WITH BIMZELX NAVIGATETM
BIMZELX NAVIGATETM helps make the treatment journey smooth from the start for your patients.
WANT MORE INFORMATION ABOUT BIMZELX?